|
They can also form complexes with metals, such as cobalt and nickel, to form hard alloys used in industry. Carbide ions are important in many chemical processes, including reactions that involve the transfer of electrons between atoms or molecules. It is composed of one carbon atom with four electrons, forming a covalent bond with the four negatively charged electrons. What Type of Ion is C4?Ĭ 4 is a carbide ion, which is an anion with a charge of 4−. Carbon also forms other monatomic ions, including C2+ and C6+, as well as polyatomic ions such as CO32. Alternatively, when it loses four electrons, it forms the carbon cation (C4+). When it gains four electrons, it forms the carbide anion (C4-). It is capable of forming both cations and anions. This is known as double-bonded carbon, and it is the reason why CO2 has a neutral formal charge overall. This is because each oxygen atom has a charge of -1, while the carbon atom has a total of 2 bonds to the oxygen atoms, meaning it has a charge of +2. It is a neutral atom, meaning that it has an equal number of protons and electrons in its nucleus, so the total positive charge of the protons and the total negative charge of the electrons cancel each other out, resulting in a net charge of zero. The Charge of Carbon: Positive or Negative?Ĭarbon is neither a negative nor a positive ion. 
In order to complete its octet, it can either lose or gain 4 electrons and forms covalent bonds by sharing these electrons with other atoms. This is because carbon has 4 electrons in its valence shell, which is its outermost shell. With that being said, further research is necessary to get a better grasp on all aspects of this important topic! Charge of Carbon In conclusion, understanding carbon’s charge ion can help us understand how this essential element interacts with other elements and forms varius compounds which are essential for life on earth. This process is known as oxidation and gives the resulting compound an overall negative charge due to the excess of negatively charged ions present in it. On the other hand, when carbon forms an ionic compound with another element such as silicon, it gains four electrons forming carbide (C4 -) and silicide (Si4 -) anions. This makes sense since oxygen needs two more electrons to fill up its octet and carbon can provide them through covalent bonding. Carbon will have +2 charge, while each oxygen atom will have -1. When two oxygen atoms are single bonded to carbon, then the formal charge of each atom will be 0. Neutral carbon-12 (or any carbon atom) has 6 electrons with a total negative charge of 6e- orbiting a nucleus with a total positive charge of 6e+, so that the total net charge is zero. 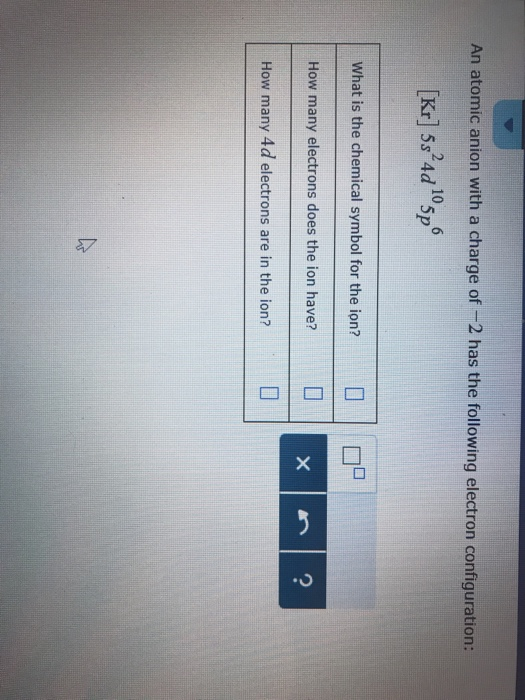
To do this, it shares its valence electrons with other atoms and forms covalent bonds. This means that it can lose or gain 4 electrons in order to complete its octet – the number of electrons needed to achieve stability. 
At a basic level, carbon has 4 electrons in its valence shell or outermost shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
